We have thousands of genes, and they are the blueprint for our growth and development, as well as controlling how our bodies function. If a particular gene is altered, then it may not work efficiently.
Genes are carried on structures called chromosomes. It is usual to have 23 pairs of chromosomes that are numbered in pairs from pair 1 to pair 22, plus one pair of sex chromosomes: XX for a female and XY for a male.
Each gene is a template for making a protein needed for the body to function. A child will inherit one set of chromosomes from their mother in the egg, and one set from their father in the sperm. Therefore, we each have two copies of each gene, one of which is inherited from each parent.
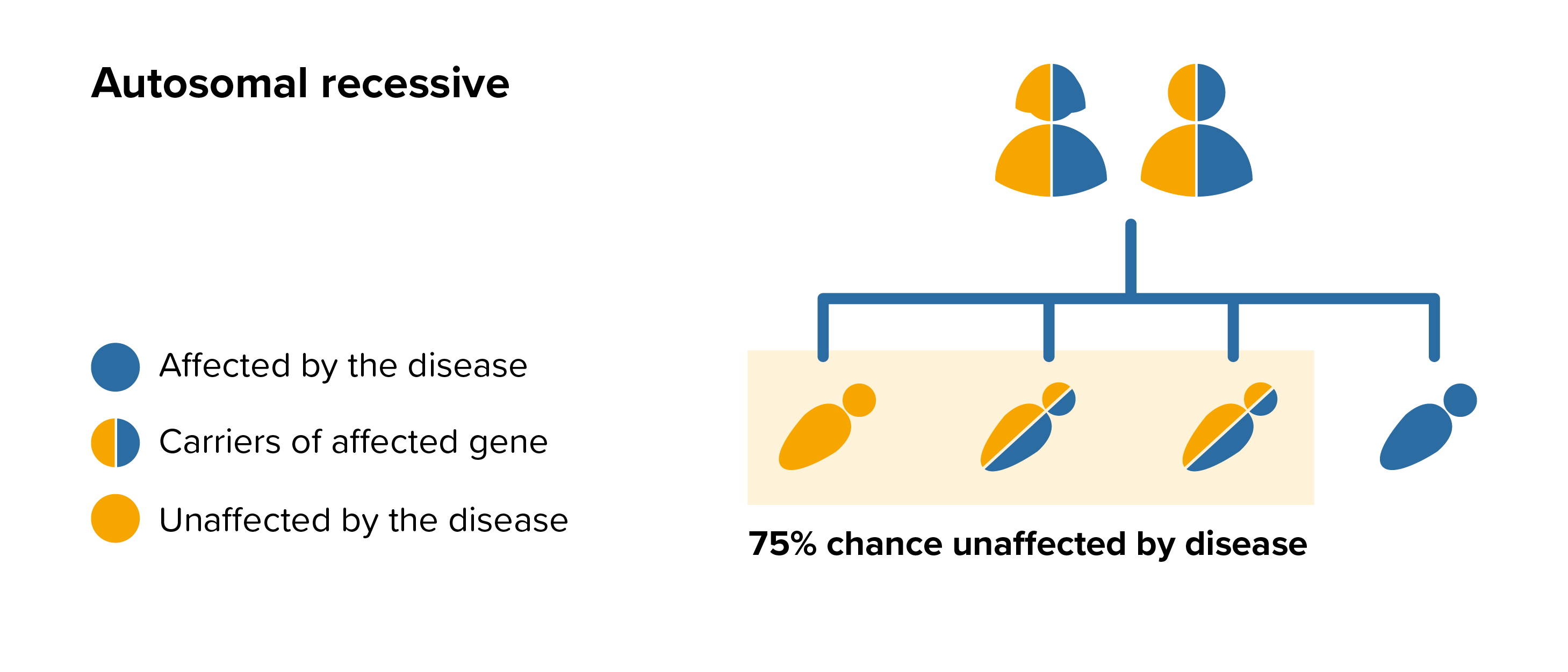
In a person with MPS III, both copies of the associated gene have genetic alterations (mutations). This is an autosomal recessive inheritance pattern. Both parents each carry one copy of the altered gene, but they do not show signs and symptoms of the disease. This is known as being a carrier. Brothers and sisters of a person affected by MPS III might also be carriers of the condition and they may wish to be referred to their local genetic department about the potential risks in future pregnancies.
 Genetic counselling
Genetic counselling
MPS III is a genetically inherited condition and there is a risk of recurrence in future pregnancies for a couple with an affected child. Therefore, parents of children with MPS III should be referred to a genetic counsellor. The counsellor will provide non-directive advice on reproductive choices, the risk to close relatives, and will suggest whether the wider family should be informed. In order to access genetic counselling, you can request a referral from either your specialist centre or your GP. Genetic services are provided by regional genetic centres.
More information about inheritance is available in our publication.
Testing for MPS III Sanfilippo in pregnancy
It is important to contact your doctor if you are planning to become pregnant, or as soon as you suspect that you may be pregnant, if you wish to discuss your family history and options. Both amniocentesis and chorionic villus sampling can be used to diagnose MPS III during pregnancy.
Amniocentesis involves using a needle to take a sample of fluid from around your baby. This fluid is then used to test the baby’s DNA for genetic alterations in your family. Chorionic villus sampling is similar to amniocentesis but instead involves testing a sample of cells from where the placenta attaches to the uterus.
Pre-implantation genetic testing (PGT) is an assisted fertility treatment. It may be possible to have PGT to avoid passing MPS III to the baby if you and your partner are known to be carriers of the condition. PGT involves checking the chromosomes of embryos for gene alterations before they are implanted in the womb, using IVF techniques. This is a complex process and requires referral from your regional genetics service.

 Genetic counselling
Genetic counselling








